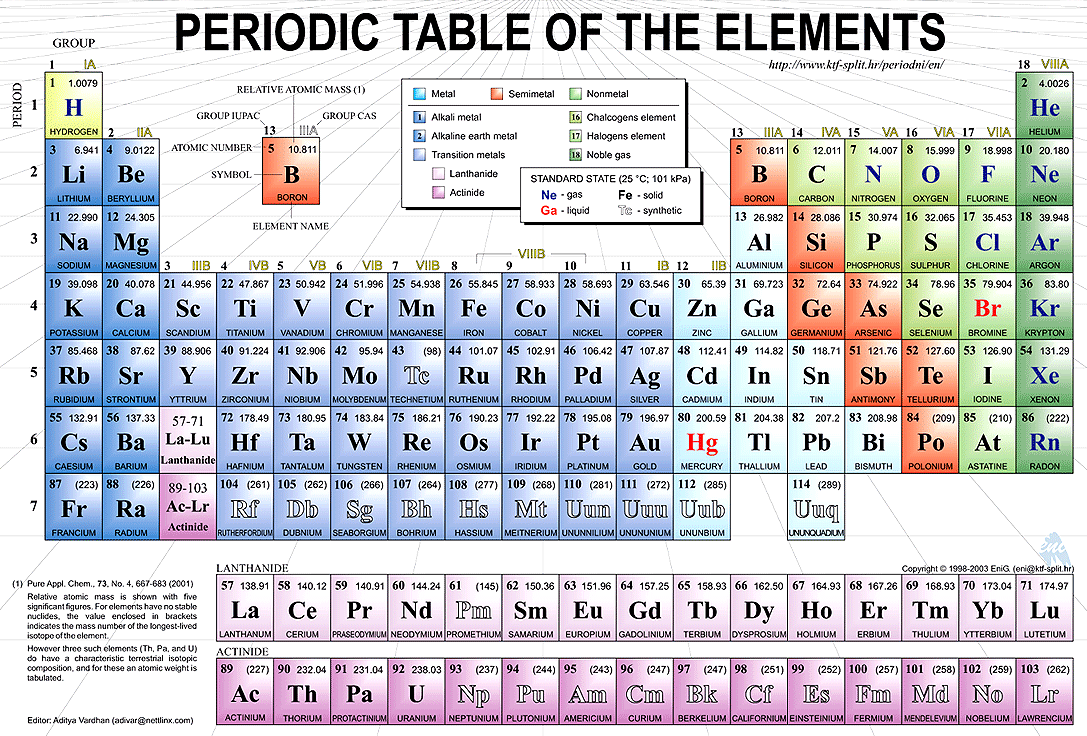
Goal 1 - The Periodic Table
Periodic Table Prelab
Go to the following website -
http.//chemicalelements.com/
Click on Alkali Metals (left bar) and answer the following questions DCN Style using word. Title this section Alkali Metals!
1. What is the group number? 2. Are these metals reactive? 3. Do these metals occur freely in nature?
4. How many electrons are in their outer shell?
5. What are the three characteristics of ALL metals?
6. Are these metals soft or hard?
7. Name the two most reactive elements in this group?
8. What happens when they are exposed to water?
Click on Alkaline Earth Metals (left bar). Title this section Alkaline Earth Metals.
9. What is the group number?
10. Are these metals reactive?
11. Do these metals occur freely in nature?
12. How many electrons are in their outer shell? _________ (Hint: It’s the same as their oxidation number or group number.)
Click on Transition Metals (left bar) . Title this section Transition metals.
13. How many elements are in this group?
14. What are the group numbers?
15. What are valence electrons?
16. Because the valence electrons are present in more than one _____________ transition metals often exhibit several common __________________.
17. Name the three elements in this family that produce a magnetic field.
Click on Other Metals (left bar). Title this section Other Metals
18. How many elements are in this group?
19. What are the group numbers?
20. How are these other metals similar to the transition metals?
21. How are these metals different than the transition metals?
22. List three physical properties of these other metals.
23. What are the oxidation numbers for this group?
Click on Metalloids to answer these questions. Title this section Metalloids.
24. Metalloids have properties of both ________ and _____________.
25. Define semiconductor.
26. Name two metalloids that are semi-conductors.
27. This property makes metalloids useful in ____________and ______________.
Click in Nonmetals to answer these questions. Title this section Nonmetals.
28. What are the group numbers?
29. List four characteristics of ALL nonmetals. ______________________________
30. What two states of matter do nonmetals exist in at room temperature?
31. The nonmetals have no _______________and do not ____________________.
32. What are the oxidation numbers of the nonmetals?
Click on the Halogens (left bar) to answer these questions. Title this section Halogens
33. What is the halogen group number?
34. Are halogens metals or nonmetals?
35. What does the term “halogen” mean?
36. How many electrons are in their outer shell?
37. What is their oxidation number?
38. What states of matter do halogens exist in at room temperature?
Click on Noble Gases (left bar) and answer these questions. Title this section Noble Gases
39. What is the group number?
40. Why were these gases considered to be inert or stable?
41. What is their oxidation number?
Click on Rare Earth Elements ( Inner Transition) (left bar) and answer these questions. Title this section Rare Earth Metals
42. How many Rare Earth elements are there?
43. Define trans-uranium.
44. The Rare Earth metals are found in groups and periods?